NYACP Board Review Question of the Week
Every other Tuesday, NYACP members are sent a Board Review Question from ACP's MKSAP 18 to test professional knowledge and help prepare for the exam. Participant totals and answer percentages are distributed on the first Thursday of the month in IM Connected, the Chapter's eNewsletter, and are also published on this page.
If you are interested in receiving these questions bi-weekly, join us as a member!
If you are a member who needs to receive the questions and newsletter via email, let us know!
June 2nd, 2026
MKSAP 19 Infectious Disease, Question 101
A 38-year-old woman is hospitalized for fever, night sweats, and a nonproductive cough of 6 weeks' duration. Medical history is significant for Crohn disease. Her only medication is ustekinumab.
On physical examination, temperature is 39.6 °C (103.3 °F), blood pressure is 100/60 mm Hg, pulse rate is 120/min, and respiration rate is 26/min. Crackles are heard in both lung fields. Abdominal examination reveals no hepatosplenomegaly.
Laboratory studies:
Hemoglobin 10 g/dL (100 g/L)
Leukocyte count 3600/µL (3.6 × 109/L)
Platelet count 109,000/µL (109 × 109/L)
Alkaline phosphatase 312 U/L
Alanine aminotransferase 51 U/L
Aspartate aminotransferase 58 U/L
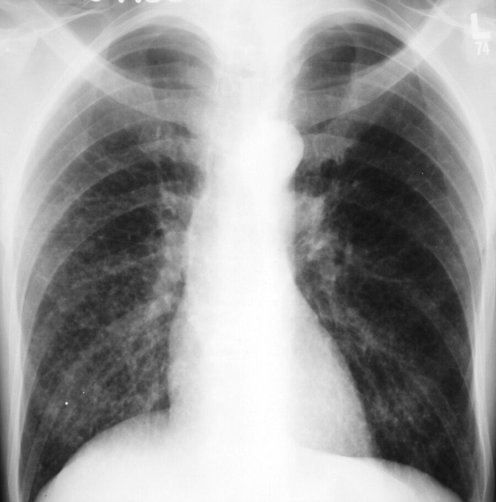
Serum and urinary Histoplasma antigen are negative. Interferon-γ release assay is negative. Chest radiograph is shown.
Which of the following is the most likely diagnosis?
A. Disseminated histoplasmosis
B. Disseminated tuberculosis
C. Streptococcus pneumoniae bacteremia
D. Subacute hypersensitivity pneumonia
Responses Received from Members (638 Responses):

The Correct Answer is: B. Disseminated tuberculosis
Educational Objective: Diagnose disseminated tuberculosis infection.
The most likely diagnosis is disseminated tuberculosis infection (Option B). This patient's use of a biologic immune modifier (ustekinumab) is a significant risk factor for tuberculosis reactivation or dissemination. The patient's interferon-γ release assay was negative, but anergy is observed more frequently among patients with miliary tuberculosis than those with pulmonary or isolated extrapulmonary involvement. Disseminated tuberculosis often involves the lungs, liver, and bone marrow. The patient's chest radiograph reveals the bilateral presence of innumerable 1- to 3-mm nodules. These findings, although not specific, are typical of tuberculosis dissemination through the vasculature or the lymphatic vessels. She also has pancytopenia, an indication of bone marrow involvement, and increased alkaline phosphatase and aminotransferase levels indicate hepatic involvement. Finally, her high fever, increased heart rate and respiration rate, and hypotension are also signs of sepsis. Although not usually in the differential diagnosis, disseminated tuberculosis can induce classic septic shock and death if not diagnosed and treated promptly and adequately.
Sepsis, pancytopenia, oral ulcerations, and hepatosplenomegaly in an immunosuppressed patient are typical of disseminated histoplasmosis (Option A). However, the Histoplasma serum and urinary antigen were negative; these tests have a sensitivity and specificity approaching 80% in immunosuppressed patients with disseminated infection, making this diagnosis less likely.
The triad of pneumococcal endocarditis, meningitis, and pneumonia (Austrian syndrome) is the result of Streptococcus pneumoniae bacteremia (Option C). However, this is an acute and rapidly progressive disease and is not compatible with this patient's 6-week course of progressive illness.
Repetitive inhalation of antigens in a sensitized patient can result in hypersensitivity pneumonitis (HP) (Option D). Patients with subacute HP have a chronic low-level exposure to antigens and experience cough, fatigue, weight loss, and shortness of breath. High-resolution CT will show micronodules and ground-glass opacities. Subacute HP does not involve the bone marrow or liver or result in sepsis.
Key Point
- Immunosuppression is a risk factor for disseminated tuberculosis, which often involves the lungs, liver, and bone marrow and may result in systemic inflammatory response syndrome, septic shock, and death.
- The interferon-γ release assay may be negative in patients with disseminated tuberculosis.
Bibliography
Sharma SK, Mohan A. Miliary Tuberculosis. Microbiol Spectr. 017;5(2):10.1128/microbiolspec.TNMI7- 0013-2016. PMID: 28281441 doi:10.1128/microbiolspec.TNMI7-0013-2016
Multiple-choice questions reprinted with permission from the American College of Physicians.
MKSAP 19. © Copyright 2021 American College of Physicians.
ACP MKSAP. © Copyright 2025 American College of Physicians. All Rights Reserved All Rights Reserved.
May 19th, 2026
MKSAP 19 Infectious Disease, Question 24
A 26-year-old woman undergoes consultation to update her vaccinations. She is an elementary school teacher. Medical history is significant for well-controlled HIV diagnosed 6 years ago. Medications are tenofovir alafenamide, emtricitabine, and dolutegravir.
The physical examination is normal.
Her CD4 cell count is 520/µL and has been stable for several years. HIV viral load is undetectable.
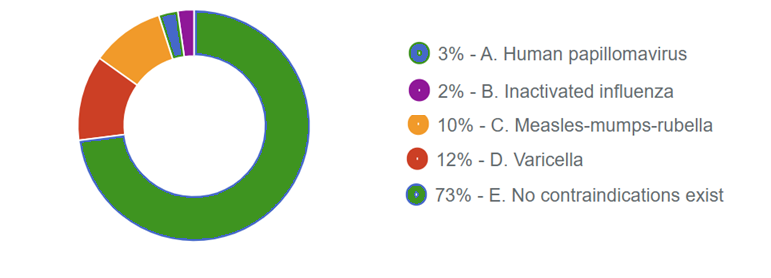
Which of the following vaccines is contraindicated in this patient?
A. Human papillomavirus
B. Inactivated influenza
C. Measles-mumps-rubella
D. Varicella
E. No contraindications exist
Responses Received from Members (707 Responses):
The Correct Answer is: E. No contraindications exist
Educational Objective: Identify appropriate vaccines for patients with HIV infection.
This patient has no contraindications to routinely administered vaccines (Option E). Live vaccines, including varicella, measles-mumps-rubella, and influenza are not recommended for patients who are severely immunocompromised. Contraindications to live vaccines include persons with HIV with CD4 cell count ≤200/µL; pregnancy or probable pregnancy within 4 weeks; immunosuppressant therapy, including high-dose glucocorticoids; leukemia, lymphoma, or other bone marrow and lymphatic system malignancies; cellular immunodeficiency; solid organ transplant recipient; and recent hematopoietic stem cell transplantation.
The live-attenuated influenza vaccine is contraindicated in patients with HIV infection regardless of CD4 cell count, but the inactivated vaccine can be given (Option B). Numerous other immunizations are recommended for all persons with HIV, including COVID-19 and pneumococcal vaccines. Patients who are not already immune or infected with hepatitis B virus should receive the hepatitis B vaccine series. Tetanus-diphtheria-pertussis, hepatitis A, and human papillomavirus (HPV) vaccinations are indicated as for the general population (Option A). The Advisory Committee on Immunization Practices (ACIP) has expanded the age indications for HPV vaccination to 45 years, recommending that the decision to vaccinate between ages 26 and 45 years be determined through shared decision making with patients. The ACIP recommends that all persons with HIV infection be vaccinated for meningococcal disease with the quadrivalent meningococcal vaccine, including boosters every 5 years.
For prevention of varicella-zoster, the inactivated recombinant vaccine is recommended for patients with HIV infection aged 19 years and older. For patients without immunity, selected live vaccines such as the measles-mumps-rubella and varicella vaccines can be given to patients with HIV infection as long as the CD4 cell count is greater than 200/µL (Option C, D).
Key Point
- Selected live vaccines, including the varicella and measles-mumps-rubella vaccines, are safe to administer to nonimmune persons with HIV infection whose CD4 cell count has consistently been greater than 200/µL.
- The live influenza vaccine is contraindicated in immunocompromised patients and those with HIV regardless of CD4 cell count.
Bibliography
Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. Updated April 17, 2020. Available at https://aidsinfo.nih.gov/guidelines/html/4/adult-and-adolescent-opportunistic- infection/0
Multiple-choice questions reprinted with permission from the American College of Physicians.
MKSAP 19. © Copyright 2021 American College of Physicians.
ACP MKSAP. © Copyright 2025 American College of Physicians. All Rights Reserved All Rights Reserved.